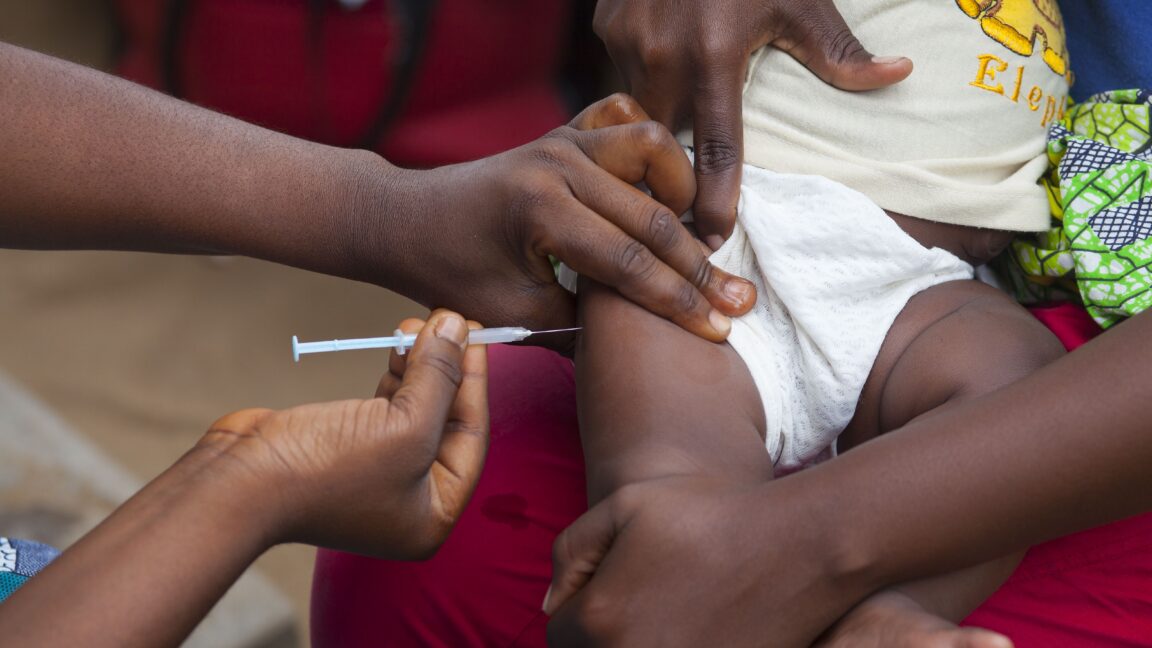
The World Health Organization (WHO) has issued an unprecedented condemnation of a US-funded clinical trial for a neonatal vaccine, labeling its design and execution as "ethically unacceptable." The trial, conducted across several low-income countries, aimed to test the efficacy of a new vaccine formulation in newborns within their first 72 hours of life. According to the WHO report, the study suffered from serious flaws in the informed consent process and failed to meet international standards for research ethics, particularly those protecting the most vulnerable populations.
The context of this trial lies within global efforts to reduce neonatal mortality, which remains a public health challenge in regions with fragile health systems. While vaccine development for this age group is a legitimate scientific priority, the WHO emphasizes that urgency cannot justify omitting fundamental ethical safeguards. The report details that many participating families came from communities with low literacy rates and limited access to medical information, compromising their ability to fully understand the trial's risks and benefits. Furthermore, researchers did not provide adequate standard care alternatives, a direct violation of the Declaration of Helsinki.
Relevant data indicate the trial, which began three years ago, involved approximately 2,500 newborns across three countries. The vaccine in question was intended to prevent a severe bacterial infection with a high mortality rate in infants. However, the WHO notes that the rate of recorded serious adverse events was significantly higher than anticipated in the initial protocol, and this data was not communicated transparently and promptly to local regulatory authorities. Funding came largely from a US federal agency dedicated to biomedical research, raising questions about the oversight mechanisms for funds allocated to international studies.
In statements gathered by the WHO ethics committee, Dr. Anya Sharma, director of the Department of Standards and Research for Health, stated, "The protection of participants, especially newborns, must be the cornerstone of any research. This trial failed in that primary duty. The exploitation of vulnerable communities in the name of scientific advancement is a stain on the conscience of global health." Meanwhile, the research institute responsible for the trial issued a statement defending its protocols but announced an independent internal review following the criticism.
The impact of this controversy is multifaceted. Firstly, it erodes community trust in health systems and medical research, a particularly damaging effect in regions where distrust of external interventions is already high. Secondly, it raises serious questions about the governance of clinical trials sponsored by wealthy nations and conducted in poor countries, a power imbalance that bodies like the WHO have been trying to correct for years. Finally, it could slow the legitimate development of urgently needed neonatal vaccines, as donors and regulators might impose additional barriers out of an excess of caution.
In conclusion, the case exposes a systemic failure in applying global ethical frameworks. The WHO has called for a moratorium on enrolling new participants in similar trials until reinforced and binding oversight mechanisms are established. The international community must reflect on how to balance medical innovation with social justice, ensuring that the search for solutions for the most vulnerable does not paradoxically turn them into victims of the process. The integrity of science depends on the ethics with which it is practiced, and this episode serves as a somber reminder of the consequences of ignoring that fundamental principle.